Novel Food Sublicense
Novel Food Sublicense - How To Apply
To acquire a sub-license for your company, please:
- Establish your total CBD related yearly turnover
- Fill out our sub-license contract
- Print and sign
- Post a physical copy to our Dutch office
- Pay the first installment into our bank account
As you may be aware, Novel Food guidelines are expected to be enforced this year. In the UK, this process will start after March 31st. The EU member states will follow later this year.
To make sure your products can stay on the market legally, we are offering sub-licensing of our novel food application, valid in both the EU as well as the UK (EFSA and FSA).
The sub-license makes you a participant in the EIHA Projects Novel Food consortium. The cost of your sublicence will depend on your total CBD related revenue.
The four product classes that are covered are:
- Raw ( Non Decarboxylated, Full Spectrum)
- Regular (Decarboxylated Full Spectrum)
- Golden (Broad Spectrum extracts , Distillates) (Not covered in initial UK and EU application)
- Isolated (99% Pure CBD)
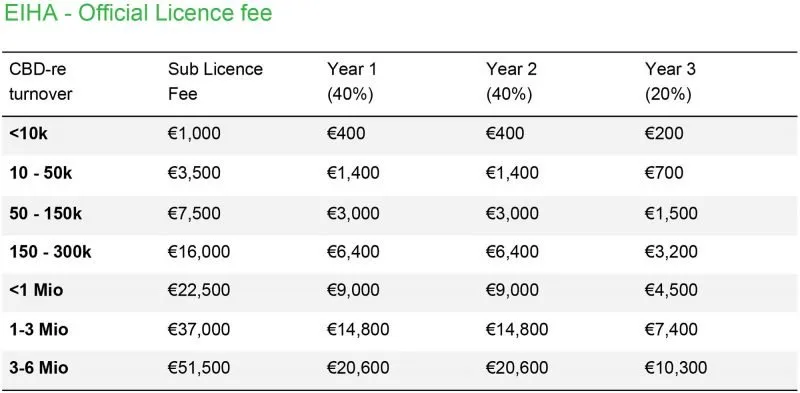
Original Pricing for clients before August 2021
Frequently asked questions
Novel Food Guidelines are a set of guidelines about what kinds of products are safe to sell as foods. In Europe, a food is considered novel, if it wasn’t regularly used for human consumption to a significant degree within the EU before 15 May 1997.
If a new product is discovered, or imported from elsewhere in the world, the business that sells it must prove that the product is safe for human consumption.
The designation of CBD under the novel food guidelines has changed several times in the last year. Currently, all food stuffs containing significant amounts of cannabinoids are deemed novel in food.
Once it is proven that a food is safe for human consumption, the EFSA will grant it market authorisation. This is the goal of the EIHA projects GmbH consortium. Such market authorisation will benefit our clients through the sub-license we are currently signing into.
The bracket of payment you fall in, is determined by your historical revenue with ourselves (the partner). If you are new, you start in the lowest bracket (under 10K).
If you bought products from us last year, the total number determines the bracket you fall into.
If you have bought with us for the past 2 years, we average your purchases with us over those 2 years.
Next year, we include your performance this year to re-calculate this average.
In year one, you pay 40% of the bracket you fall in.
In year two, you pay 40% of the bracket you fall in by then.
In year three, you pay 20% of the bracket you fall in by then.
Your CBD related revenue in this case, is just the total amount that you have ordered with us.
If you haven’t ordered yet, the revenue is 0 and you fall in the lowest bracket.
If you started ordering last year, the year total will be your average.
If you also ordered during 2019, then the years 2019 and 2020 will be added and divided by two.
No, you need to print and sign a physical copy of the contract and post it to our office address in Holland.
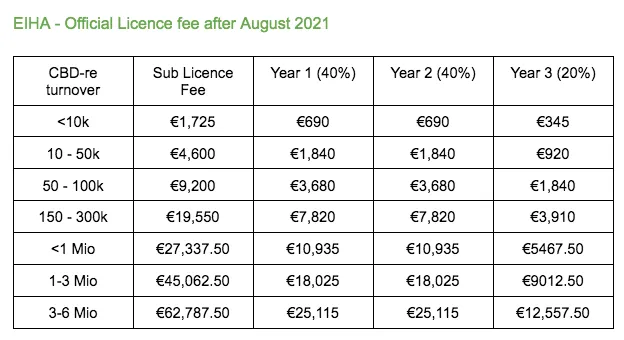
Yes. The later you join in the process, the more it will cost you. We will publish the fees with the correct increase once they are decided.
You will need a separate sublicence for every EIHA partner you purchase from.
The sub-license is only valid for the products that we deliver consumer-ready. If you buy the basic ingredient from us, this does not automatically provide a license for brands that are made with that ingredient.
If you purchase extracts and develop products for other brands, the best thing to do is become a primary EIHA member, so you can sub-license to your own clients as well.
This means you will miss the UK deadline for the 31st of March. This may have consequences for your ability to market inside the UK. In the EU, such deadline is not yet known.
There is no hard deadline yet. Keep in mind that the longer you wait, at some point the sub licences will become more expensive.
The EIHA consortium intends to get all of the 4 product categories authorized:
1 raw (unheated full spectrum extract)
2 regular (decarboxylated full spectrum extract)
3 golden (re-combined fractures or distillate)
4 isolate (99% CBD)
It is possible that not all categories will get validated simultaneously, please follow us as we will keep updating on our website.
We can add flavours to certain formulations, this has no influence to their novel food status.
When you sign into a sub-license, this covers all products, with your brand, that we supply to you.
Our consortium is using hempseed oil as a basis for all applications. We will learn later by the EFSA if they will require additional stability testing for MCT based formulas.
If they decide that such additional stability tests are required, CBD Oil Europe considers to organise and pay for those.
You are signing into an agreement with EIHA projects GmbH, so that if this effort is successful, you may benefit to the exclusion of other actors.
CBD Oil Europe is not in control of this process, and cannot guarantee a successful outcome.
It is possible that the consortium will be unsuccessful. In such case, no refunds will be issued, as stipulated in the contract.
The sublicense is made so you can use your own branding, as long as we supply the products.
Yes, this is determined only by the revenue you have placed with ourselves.
Let's talk CBD!
Get in touch and talk to one of our experts to discuss your needs. We will support through the entire process from sourcing to final distribution.